Our Blogs
Explore Real-World Product & Platform Engineering Insights



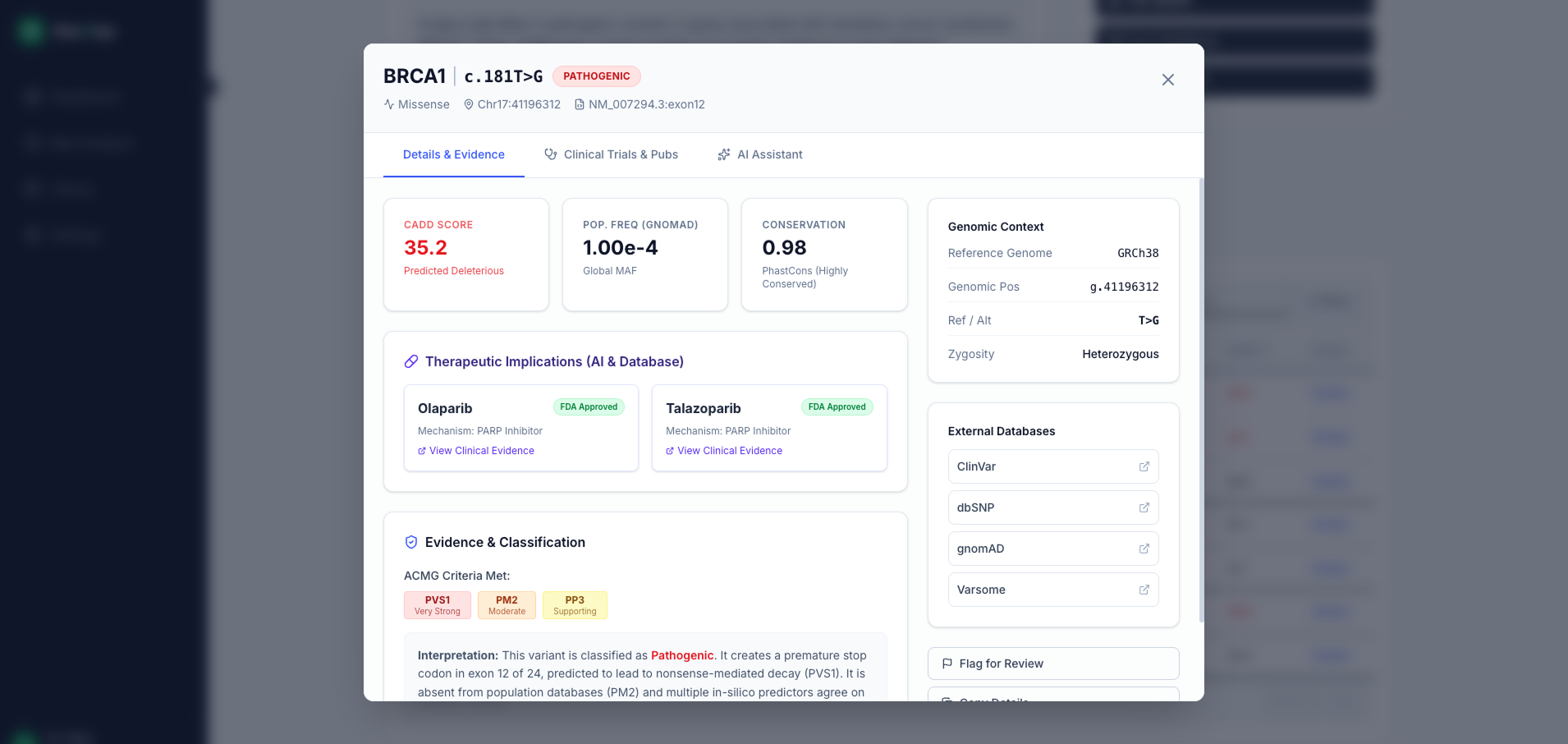
Variant Knowledge Management System (KMS) for Genomics Labs: Replace Spreadsheets Before Your Next CAP Audit
Spreadsheets can’t handle VUS reclassification, audit trails, or CAP compliance. Discover how leading genomics labs are building Variant KMS platforms to scale interpretation, reduce risk, and pass audits.

Healthcare Software Modernization: The CTO's Playbook for Replacing Legacy Clinical Systems Without Disrupting Patient Care
Here is the scenario that lands in a CTO's inbox more often than any other right now: a vendor sends an end-of-support notice for a system the clinical operations team depends on.

Why Clinical Genomics Pipelines Fail When They Move from Research to Production.
In May 2024, the FDA issued a final rule that would have required virtually every clinical genomics laboratory in the United States to seek regulatory clearance for its NGS pipelines as medical devices. A federal court vacated it in March 2025.

The ROI of Automating Sample-to-Report Workflows: A Decision-Maker’s Guide for Precision Medicine Platforms
Learn how automating sample-to-report workflows improves turnaround time, compliance, and cost control in genomics and precision medicine platforms. A decision-maker’s guide with real benchmarks and risk analysis.

LIMS Modernization 2026: A Complete Step-by-Step Guide for Genomics & Clinical Labs
Modernize your legacy LIMS without disrupting lab operations. A detailed guide for genomics, diagnostics, and clinical labs, including architecture, compliance, migration frameworks, and real-world implementation steps.


Sep 1, 2024

3 min read
LIMS Modernization 2026: A Complete Step-by-Step Guide for Genomics & Clinical Labs
Modernize your legacy LIMS without disrupting lab operations. A detailed guide for genomics, diagnostics, and clinical labs, including architecture, compliance, migration frameworks, and real-world implementation steps.


Sep 1, 2024

4 min read
Build vs Buy - Genomics Software in 2026: What Leaders Must Know
Discover the true cost of building vs. buying genomics software in 2026 - a complete decision framework for LIMS, pipelines, CDSS, and precision medicine platforms.


Sep 1, 2024

5 min read
How to Choose the Right Genomics Software Development Partner: 12 Non-Negotiable Criteria
Learn the 12 must-have criteria for choosing a genomics software development partner, from regulatory expertise to scalable cloud architecture & LIMS integration.
%201.jpg)

Sep 1, 2024

3 min read
HIPAA-Ready Genomics Platforms: Key Development Gaps & How to Fix Them
Build a truly HIPAA-compliant genomics platform. Learn the hidden risks teams overlook in architecture, pipelines, EHR/LIMS integration, security, and best practices.









.png)
.jpg)













%201.jpg)