
Clinical diagnostics is undergoing a fundamental shift. As genomics becomes embedded in oncology, rare disease diagnosis, pharmacogenomics, and inherited condition screening, the central challenge is no longer whether genomic data can be generated. It is whether that data can be operationalized reliably, integrated into clinical systems, and sustained under regulatory scrutiny at scale.
Genomics data engineering services play a decisive role in this transition. They sit between sequencing technologies, bioinformatics pipelines, clinical interpretation workflows, and downstream systems such as LIMS, EHRs, and reporting platforms. When this layer is weak or fragmented, clinical diagnostics platforms struggle to scale, reproduce results, or meet compliance expectations, regardless of scientific sophistication.
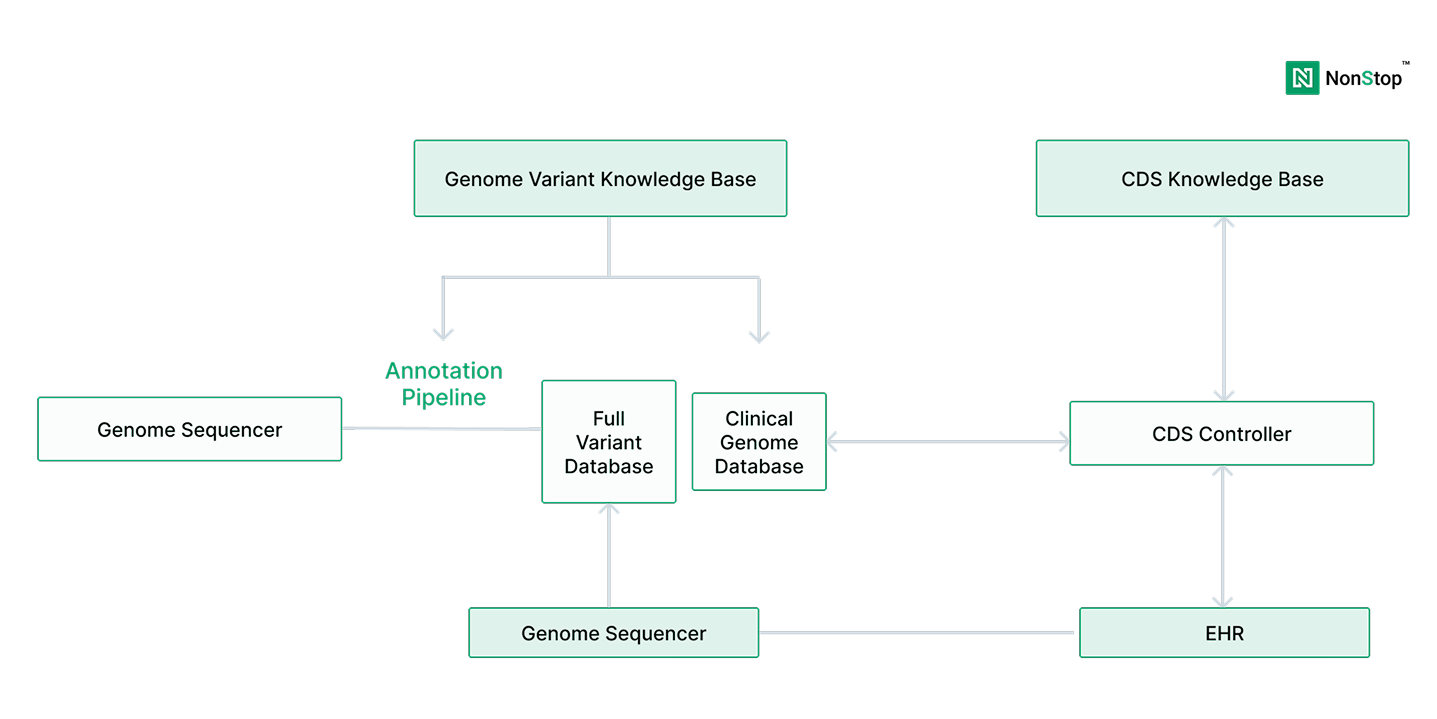
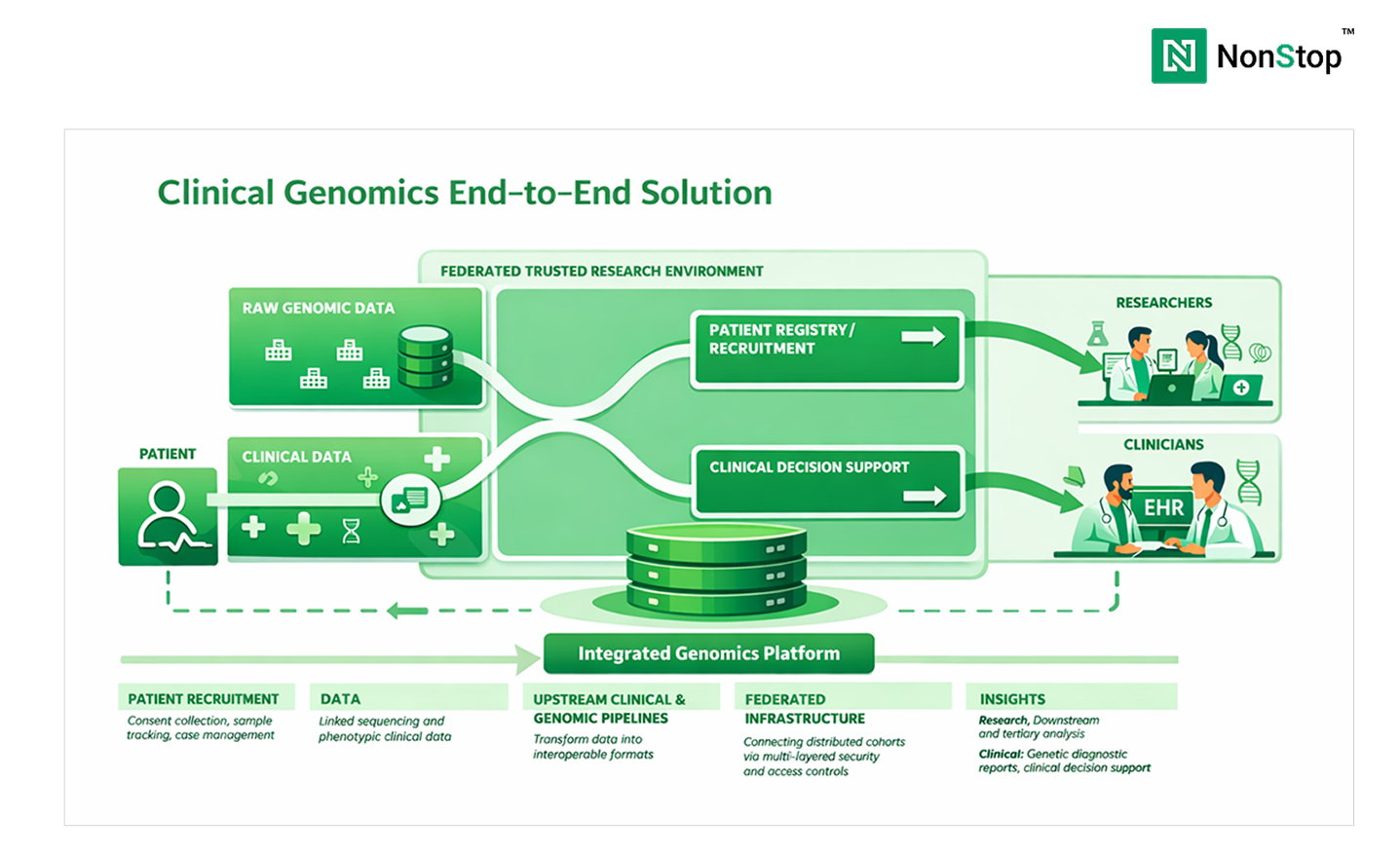
According to the National Human Genome Research Institute (NHGRI), the cost of sequencing a human genome has fallen from over $100 million in the early 2000s to under $1,000 today. As a result, genomic sequencing has moved from research settings into routine clinical diagnostics.
However, sequencing alone does not deliver clinical value. Diagnostics organizations depend on interpretation, traceability, reproducibility, and integration with care workflows. These requirements are not solved by faster sequencers or better variant callers - they are solved by platforms.
Research published in Nature Genetics and The New England Journal of Medicine repeatedly highlights a consistent gap: while variant detection accuracy has improved significantly, many labs struggle with longitudinal data reuse, interpretation consistency, and audit-ready workflows.
Genomics data engineering services address this gap by transforming analytical outputs into durable clinical systems.
A common mistake in diagnostics organizations is assuming that robust bioinformatics pipelines equate to production readiness. They do not.
Bioinformatics pipelines are designed to answer analytical questions, including variant calling, annotation, filtering, and prioritization. Clinical diagnostics platforms must answer operational ones.A common misconception is that production readiness depends primarily on selecting the right tools - a workflow engine, a data warehouse, or an AI framework.
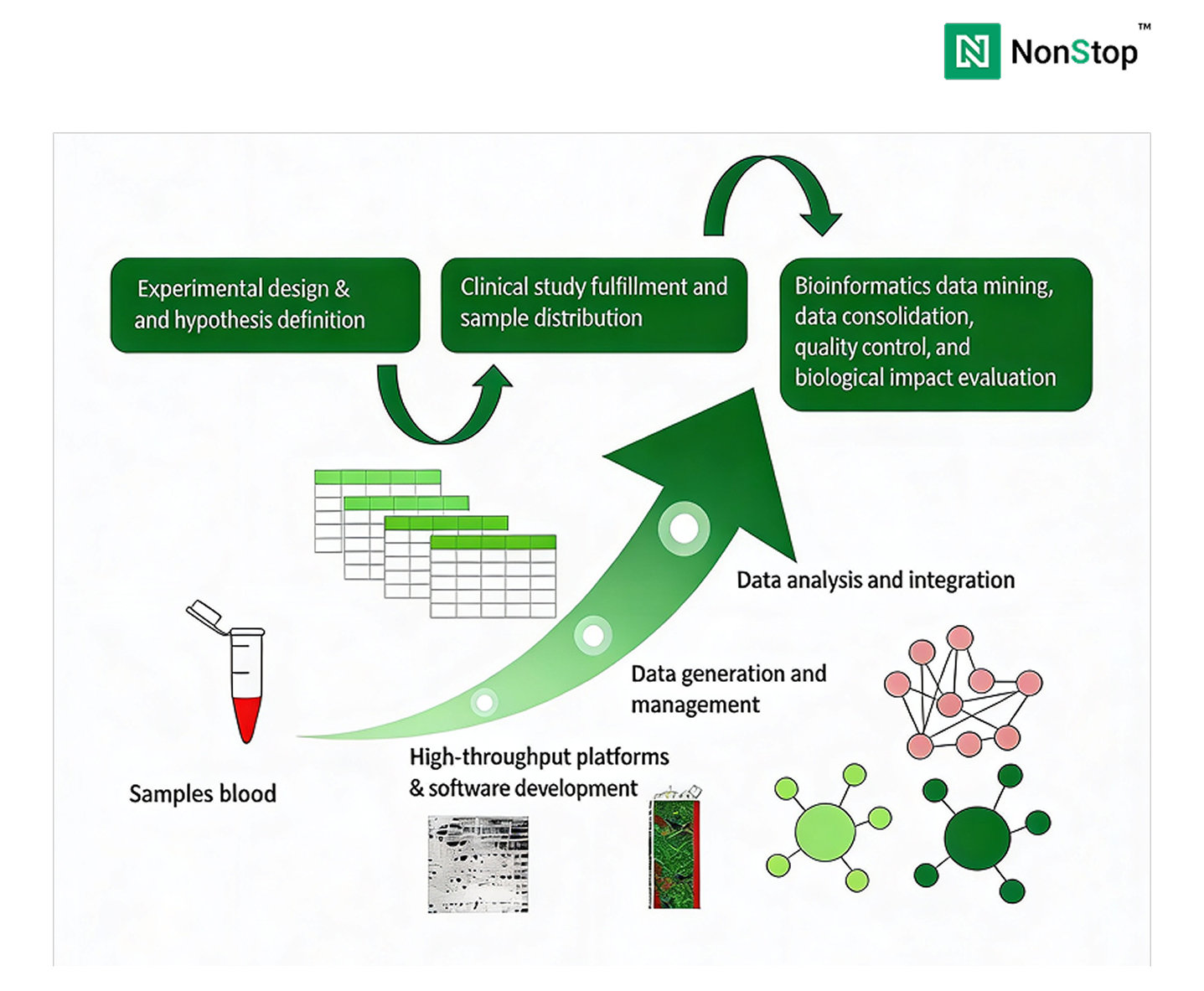
A production-grade genomics data platform must support:
Without this foundation, pipelines remain fragile, even when they produce accurate results.
Many diagnostics teams realize too late that scaling clinical genomics is less about adding new workflows and more about stabilizing the platform underneath them.
Talk to our Platform Experts
Genomic data is uniquely demanding in both scale and longevity. A single whole-genome sequence typically produces 100–200 GB of raw and intermediate data, depending on coverage and pipeline design.
At the clinical scale, programs quickly accumulate petabytes of data, even before accounting for annotations, derived features, and repeated re-analysis.Re-analysis is not an edge case. The American College of Medical Genetics and Genomics (ACMG) has documented that variant classifications change over time as evidence evolves.
This makes reproducibility, lineage, and version control clinical requirements, not optional enhancements.Without strong genomics data engineering:
Production-ready platforms treat genomic and clinical data as long-lived assets, not transient outputs.
Early genomics diagnostics initiatives often succeed. Pipelines run.
Reports are delivered. Stakeholders gain confidence.But when these initiatives are expected to run continuously, supporting targeted clinical programs, scaling across defined patient populations, and operating under regulatory scrutiny, structural weaknesses begin to surface.
1. Definition of PICOS:
2. Precise localization of clinical problem:
1. Source of data system
2. Data deduplication
3. Data evaluation
1. Assessment of impact of missing value/imputation
2. Data transformation
3. Data standardization
1. Clinical significance of features
2. Distribution of features
3. Contribution of the feature to the outcome
4. Method of feature selection
1. Determining the optimal model metrics
2. Model training
3. Method for model selection
1. Accuracy of model
2. Clinical significance
Common failure modes include:
Across healthcare and life sciences, studies consistently show that 60–70% of data and AI initiatives fail to reach sustained production use, with primary causes tied to data readiness, governance, and operational complexity rather than analytical quality.
At this stage, the issue becomes a leadership concern, rooted in platform design, ownership, and long-term strategy.
Cloud cost is one of the least predictable aspects of scaling diagnostics platforms. FinOps Foundation guidance and large-scale cloud architecture studies show that, in data-intensive platforms, compute is often not the dominant long-term cost driver.
Instead, storage growth, retries, inefficient orchestration, and data movement can account for 30–50% of total platform cost over time.
While spot or preemptible compute can reduce raw compute costs by 70–90%, these savings only materialize when pipelines are engineered for failure tolerance, observability, and reproducibility.
AI is increasingly central to clinical diagnostics strategies, but operational success remains limited. Gartner and McKinsey report that only 20–30% of AI initiatives reach sustained production use across industries.
In healthcare and genomics, the blockers are rarely model accuracy. They are operational:
Regulatory guidance increasingly emphasizes traceability and defensibility for AI-assisted decision support. Without production-grade data engineering, AI outputs remain difficult to trust and even harder to scale responsibly.
Genomics data engineering provides the foundation that AI systems depend on.
Before scaling clinical diagnostics platforms, leaders should pressure-test readiness across five dimensions:
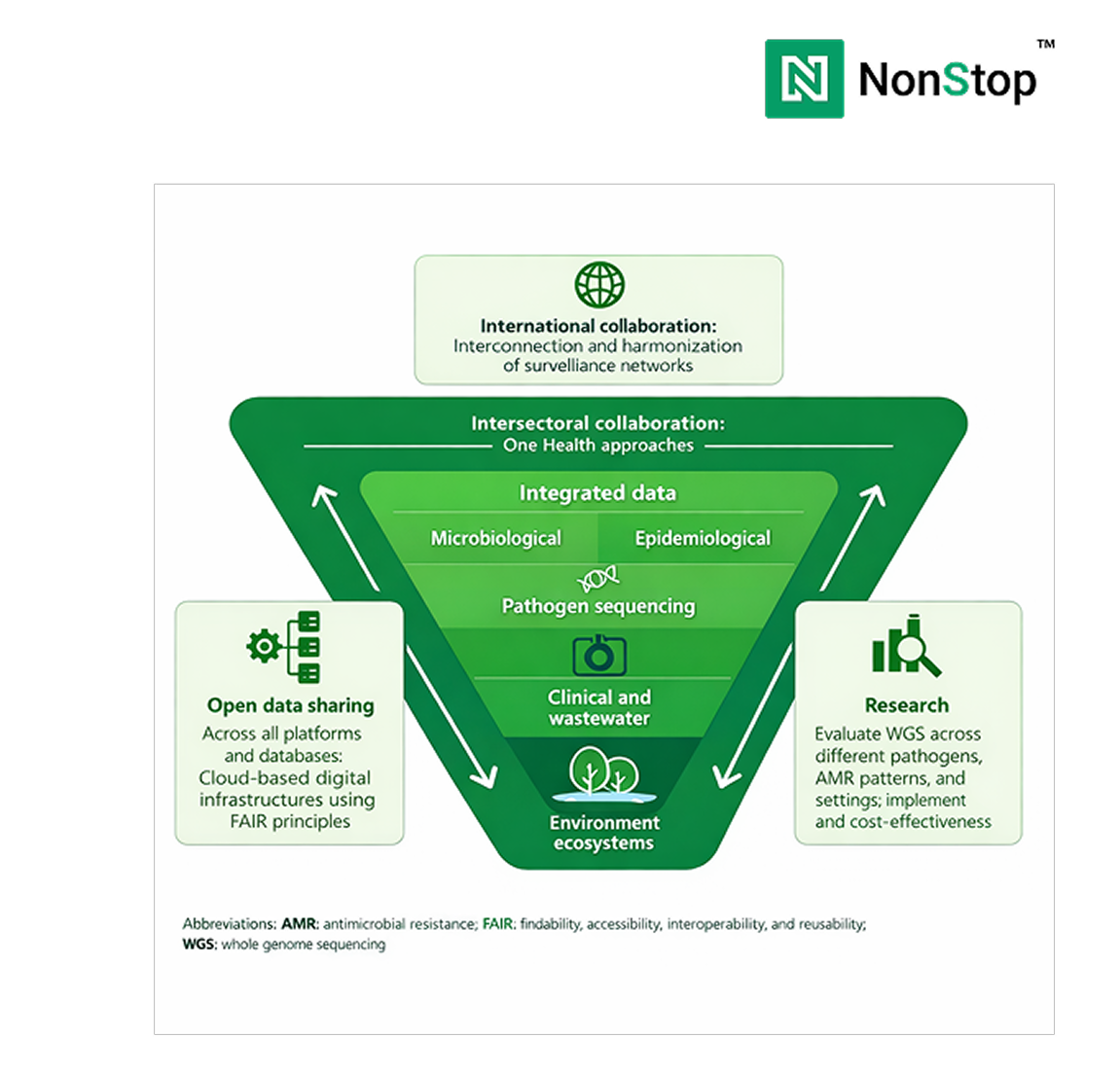
Gaps in any of these areas tend to surface later as cost overruns, compliance risk, or operational fragility.
Organizations typically engage NonStop when genomics diagnostics programs reach a critical inflection point, when early analytical success must become dependable clinical operations.
NonStop works as an engineering and platform partner, helping teams:
The focus is not on replacing scientific expertise. It is on building systems that allow that expertise to operate reliably over time.
When genomics data engineering is done well, diagnostics organizations consistently achieve:
These outcomes compound over time, creating a durable operational advantage.
Building a modern clinical diagnostics platform is not a tooling decision. It is a systems and governance decision with long-term consequences.
The organizations that succeed are those that:
The strategic question is no longer whether genomics will shape clinical diagnostics. It is whether the platform supporting it is built to scale responsibly, operate predictably, and earn trust under pressure.
If your diagnostic platform is approaching that inflection point, a focused, pratical discussion can often clarify where operational risk and cost are likely to surface next and what to address before scale makes change far more expensive.
Talk to our Experts