
Modernizing a legacy LIMS (Laboratory Information Management System) is no longer optional for genomics and diagnostics organizations; it's a matter of operational survival.
A 2024 Deloitte survey found that 92% of genomics labs cite LIMS modernization as their top digital transformation priority, yet 63% report failed attempts or major disruptions during migration, primarily due to poor planning, compliance gaps, and lack of architectural foresight.
The reality is simple:
Yet many labs approach modernization like a simple IT upgrade. They treat it as “moving data to a new platform,” rather than rebuilding the foundation for an evolving genomic enterprise.
This is where projects fail and costs explode.
In this guide, we’ll walk through:
LIMS platforms originally emerged to manage sample tracking, lab inventory, and operational workflows. In traditional clinical labs, they handled predictable data types and linear processes. Genomics changes everything:
FASTQ, BAM/CRAM, VCF, QC reports
multi-stage computational workflows, reproducibility requirements
sequencers, bioinformatics tools, EHR systems, payer systems, reporting tool
HIPAA, CLIA, CAP, SOC 2, GDPR and genetic privacy regulations
assays, pipelines, and interpretation logic change continuously
Most legacy LIMS systems cannot adapt to these dynamics. Their data models are rigid, integration layers are brittle, and auditability is often weak.
A lab cannot “pause” operations. If your modernization effort disrupts sample tracking, instrument connectivity, or reporting, it impacts turnaround time, revenue, and patient outcomes.
If your new architecture fails a HIPAA risk assessment, SOC 2 audit, or CLIA/CAP validation, the project is not just delayed, it may become unusable in clinical environments.
Many labs modernize by purchasing a COTS LIMS and customizing it. They later discover that:
Alternatively, labs build custom systems and become dependent on a small internal engineering team. When key developers leave, the platform becomes unmaintainable.
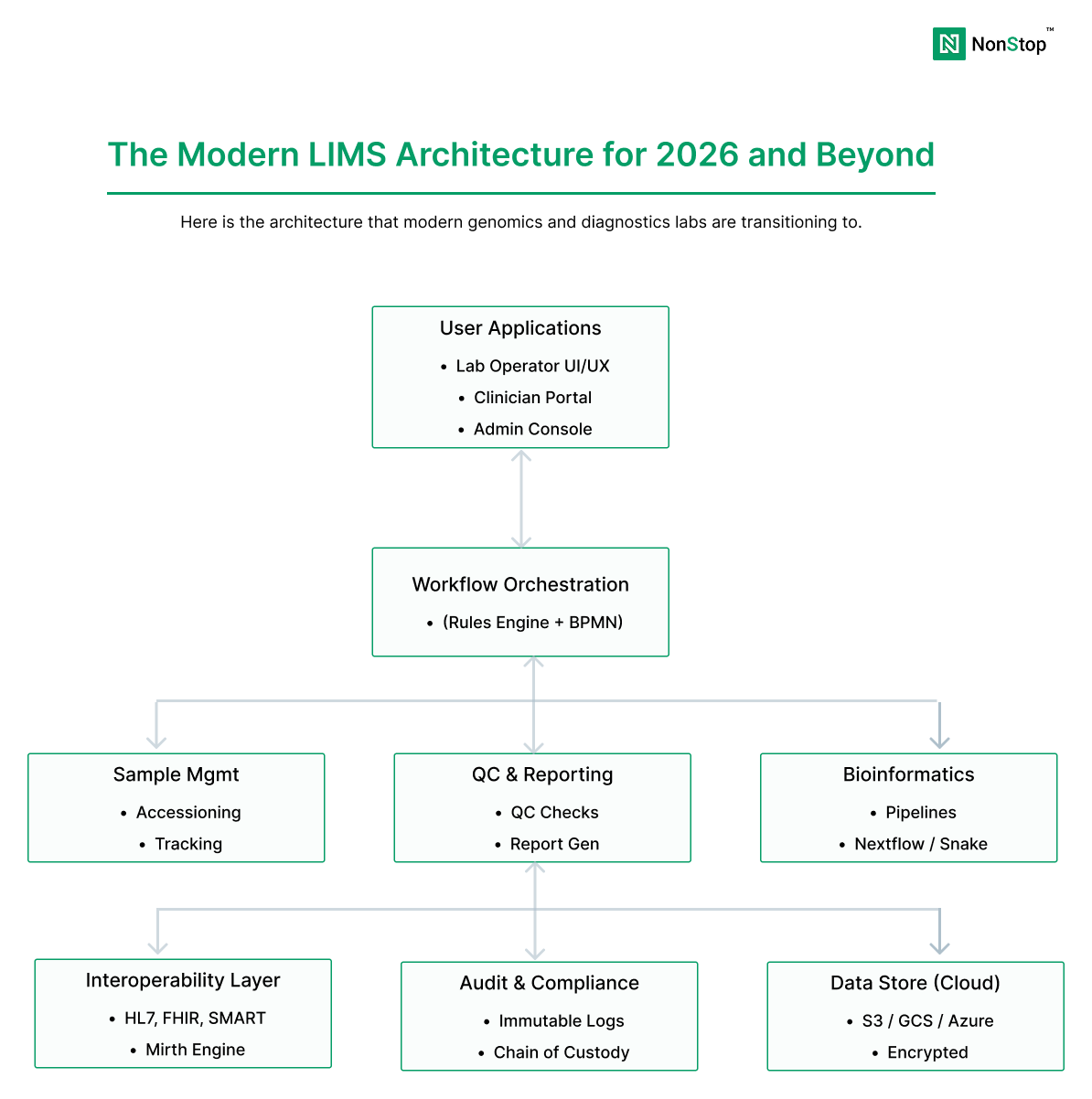
A modern LIMS platform for genomics typically separates concerns into distinct layers:
sample accessioning, QC, chain of custody, batching, instrument integration
pipeline orchestration, compute, reproducibility, outputs
annotation, scoring, clinical interpretation, report generation
EHR, billing, partner portals, CROs, data exchange standards (HL7/FHIR)
audit logs, access controls, monitoring, incident response readiness
This modular architecture allows labs to modernize incrementally, replace components without re-validating the entire system, and scale compute and storage independently.
Start by clarifying what you are modernizing and why.
Key scope questions
Most modernization failures occur because teams underestimate workflow complexity.
Document:
A genomics LIMS almost always contains PHI because sample identifiers, patient metadata, and genomic files can be re-identifiable.
Data Type
Classi
fication
Moderniz
ation Implication
Patient demog
raphics
PHI
Requires strict access control and audit logs
FASTQ/
BAM/
CRAM
PHI / Identifiable
Must be isolated in secure processing zones
VCF + clinical metadata
PHI
Requires encryption at rest/in transit; PHI-safe logs
Aggregated statistics
Non-PHI (if de-identified)
Can support analytics without PHI exposure
Most genomics organizations end up in a hybrid model: buy core utility functions, build differentiated workflows and integrations.
Modern LIMS platforms must integrate seamlessly with:
The integration layer is often where modernization either succeeds or fails.
Compliance is not a checklist you add at the end. You must architect for:
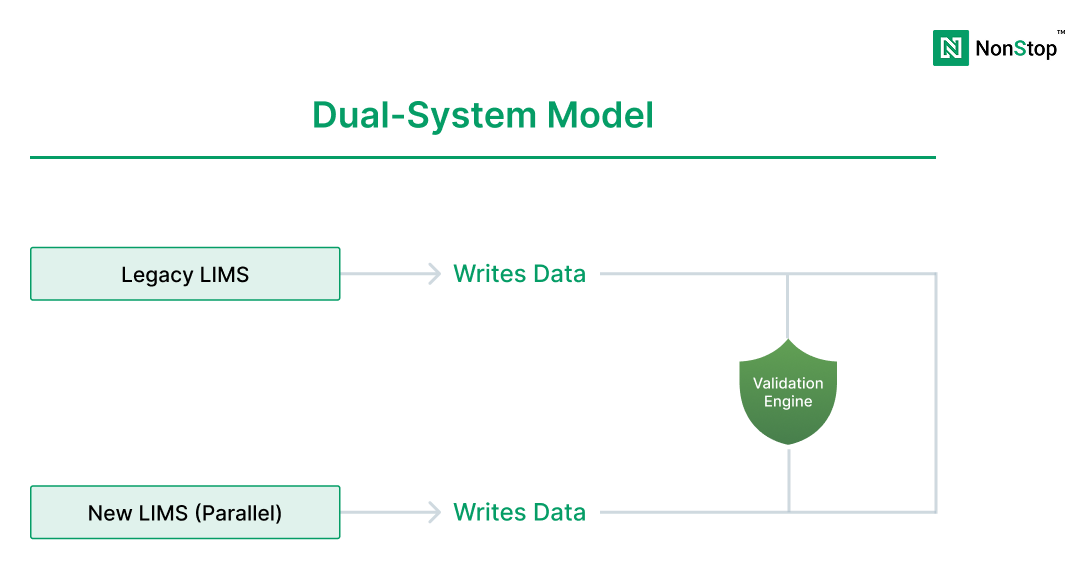
Big-bang cutovers are one of the biggest causes of lab disruption.
Preferred phased approach:
teams underestimate real lab behavior
leads to failed risk assessments and re-architecture
data quality issues surface late
locks teams into inflexible systems
integration delays dominate project timelines
causes compliance failures and deployment delays
This modular architecture allows labs to modernize incrementally, replace components without re-validating the entire system, and scale compute and storage independently.
A successful genomics LIMS modernization initiative requires more than a new UI or a database migration. It demands a full rethinking of architecture, compliance, interoperability, and operational workflows.
The labs that modernize successfully do so by: